Key figures 2025
The duration of evaluation and decision-making processes within the REACH work streams—substance evaluation, authorisation, restriction, and harmonised classification and labelling—generally exceeds one calendar year. The associated workload is therefore spread over multiple years.
The figures in this report relate to dossiers submitted by the Netherlands or on which the Netherlands has provided input. They also include dossiers for which Dutch members of the RAC and SEAC acted as (co-)rapporteurs, meaning they were responsible for drafting the opinion. RAC and SEAC members contribute to all dossiers, with or without support from other RIVM experts.
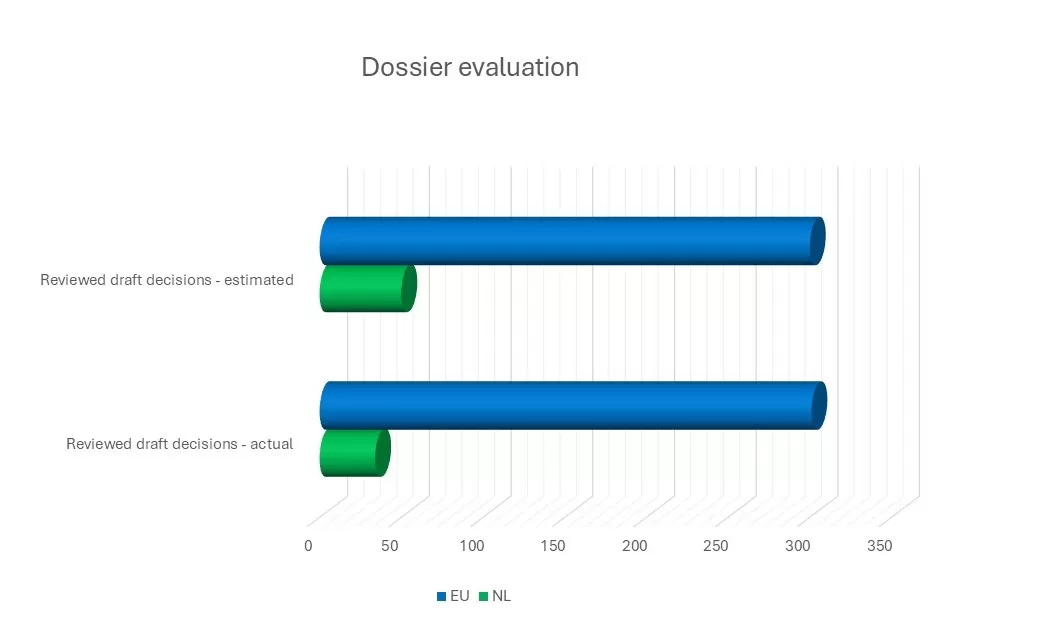
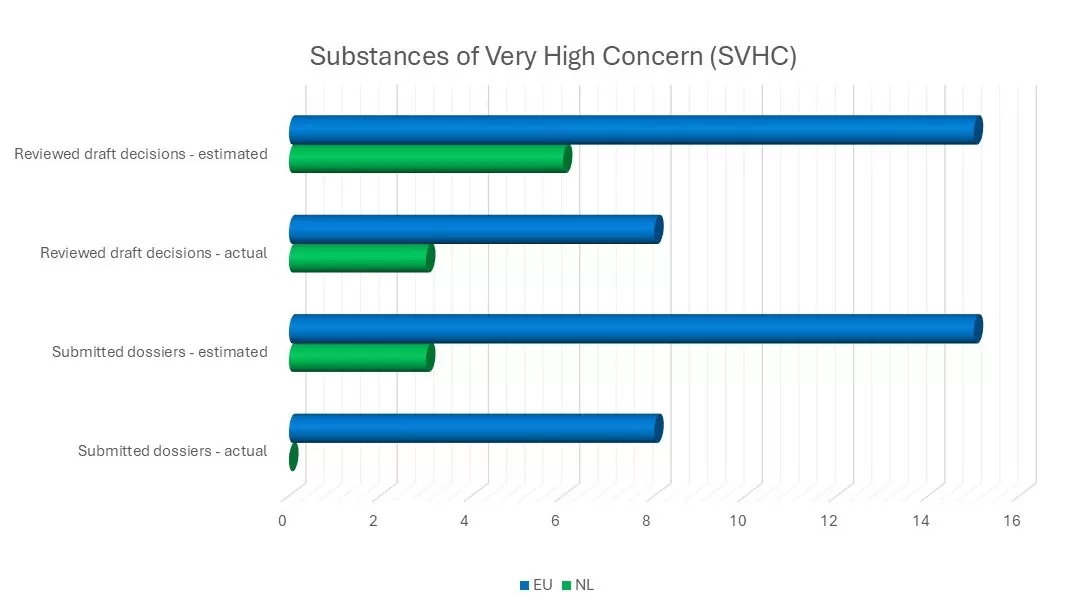
Confidence in ECHA’s dossier evaluation practice enables us to prioritise the Dutch contribution more effectively. We reviewed 34 of the total 301 proposals, resulting in four amendment proposals.
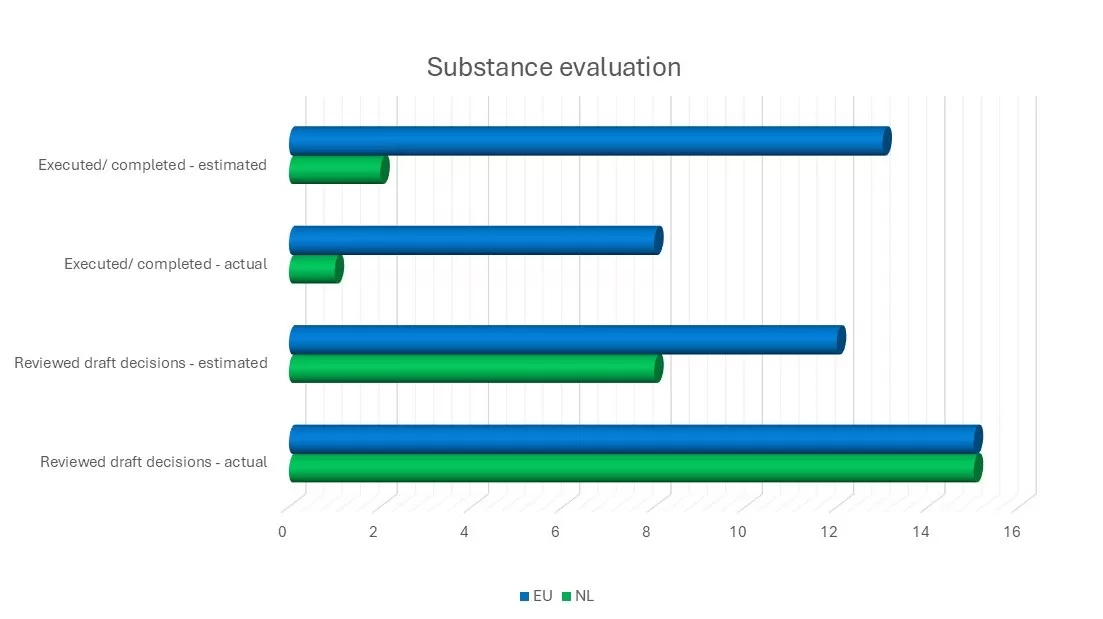
For one of the eight substances on the Community Rolling Action Plan (CoRAP), we carried out the substance evaluation on behalf of the Netherlands. In addition, we worked on eight of the 11 ongoing substance evaluations, one of which was finalised during the past year.
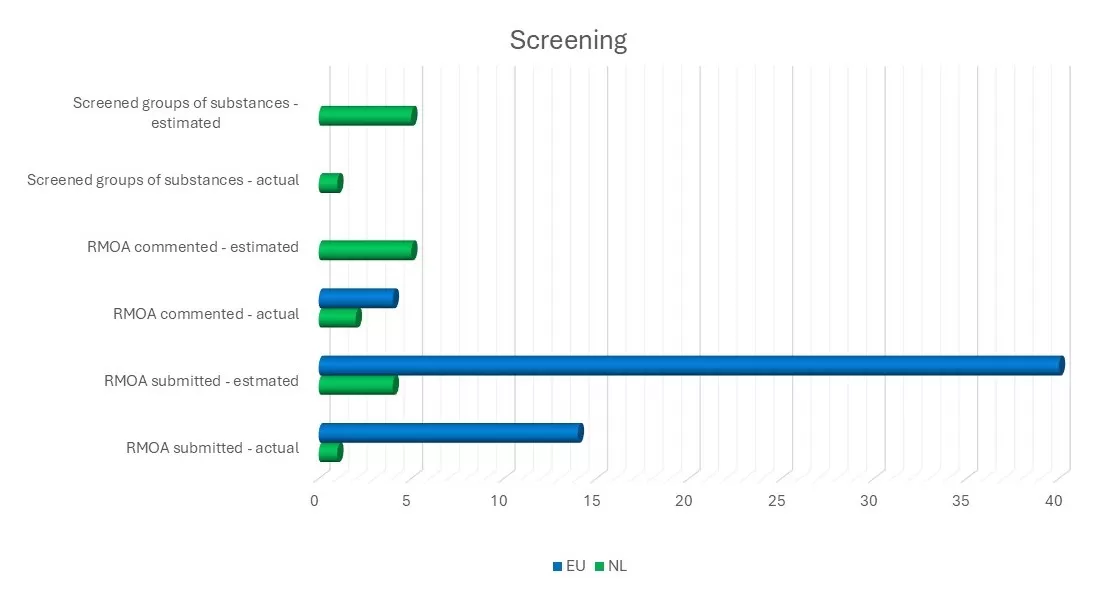
A) To identify substances that may require further regulatory action (substance evaluation, harmonised classification, authorisation or restriction), we carry out screening activities. Over the past year, ECHA shared fourteen documents on the screening of substance groups for regulatory needs (ARNs) with the Member States. Bureau REACH assessed and provided comments on one of these substance groups.
B) A Regulatory Management Options Analysis (RMOA) is used to identify and discuss the most appropriate regulatory measures with ministries, other Member States and ECHA. Bureau REACH worked on three RMOAs, one of which was completed. Four RMOAs were received from ECHA and Member States, two of which we commented on.
The Netherlands examined whether preparing an SVHC dossier for formaldehyde would provide added value in relation to existing regulations for this substance. It was ultimately concluded that SVHC identification was not necessary, as the substance is already sufficiently regulated through several specific restrictions, including under the REACH Regulation, the Toy Safety Directive and as a biocide.
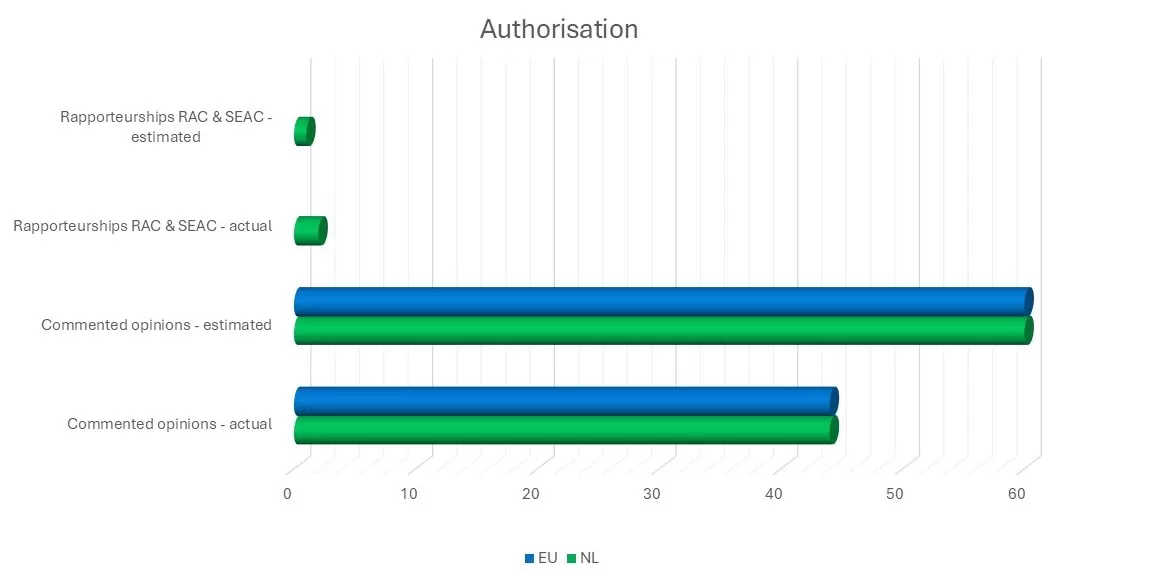
The Dutch RAC and SEAC members and their advisers contributed to all 44 authorisation applications for SVHC substances. The Dutch SEAC members acted as rapporteur for two dossiers.
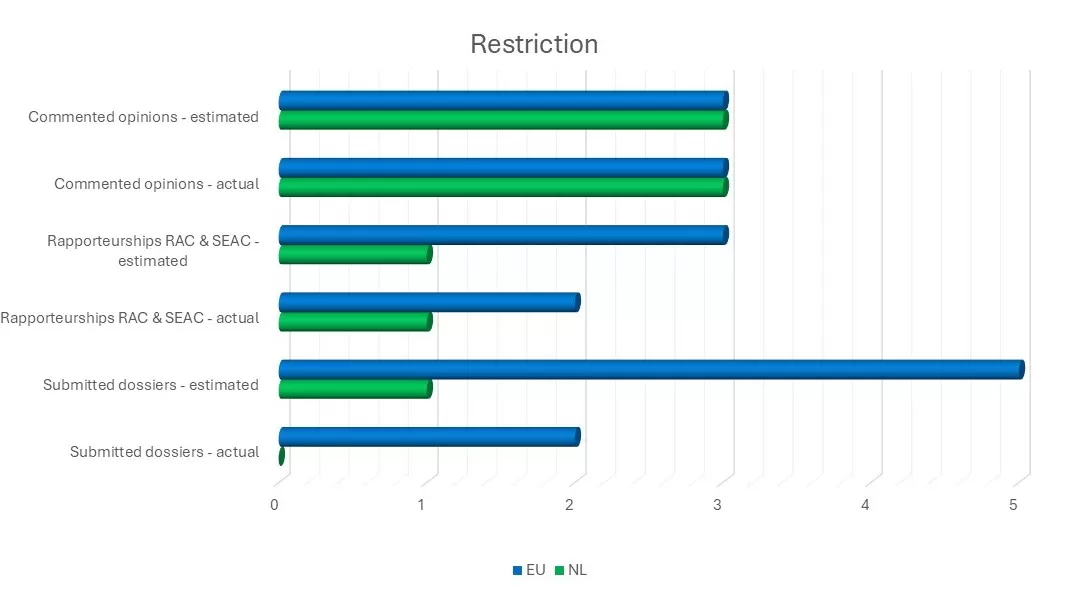
Both the PFAS restriction team and the Dutch RAC and SEAC members made substantial contributions to ECHA’s opinion-forming process on the PFAS restriction dossier.
Work on a restriction proposal for unintentionally added asbestos has now been discontinued due to anticipated limitations in enforceability.
Over the past year, in cooperation with colleagues from Austria, we initiated the development of a restriction proposal to manage the risks of toxic antioxidants (PPDs) in rubber tyres.
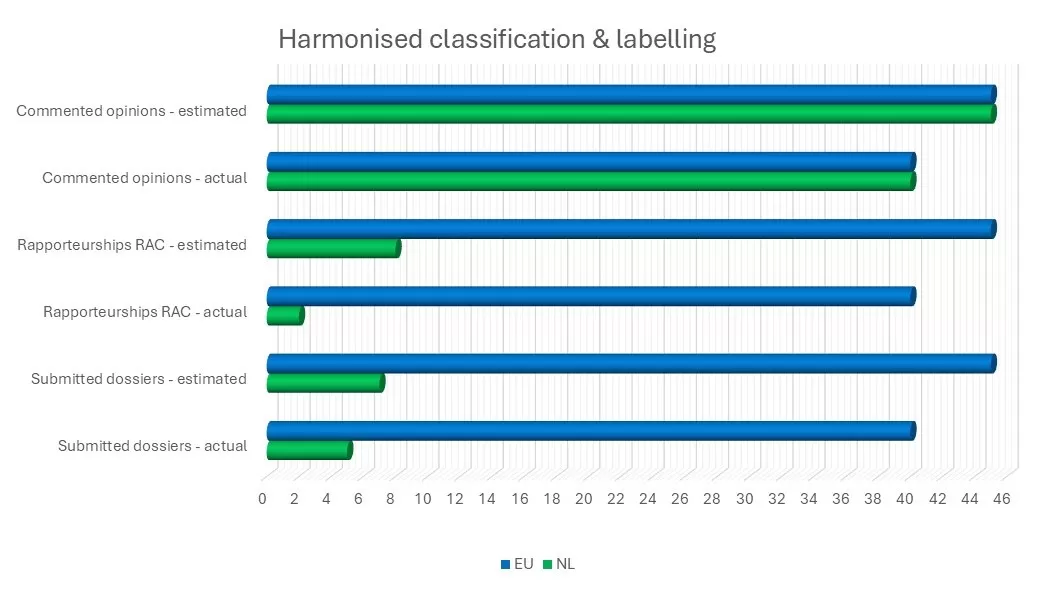
Over the past year, we submitted five harmonised classification proposals. Dutch RAC members, together with RIVM advisers, worked on two rapporteurships and provided input on all 45 opinions on harmonised classification proposals.
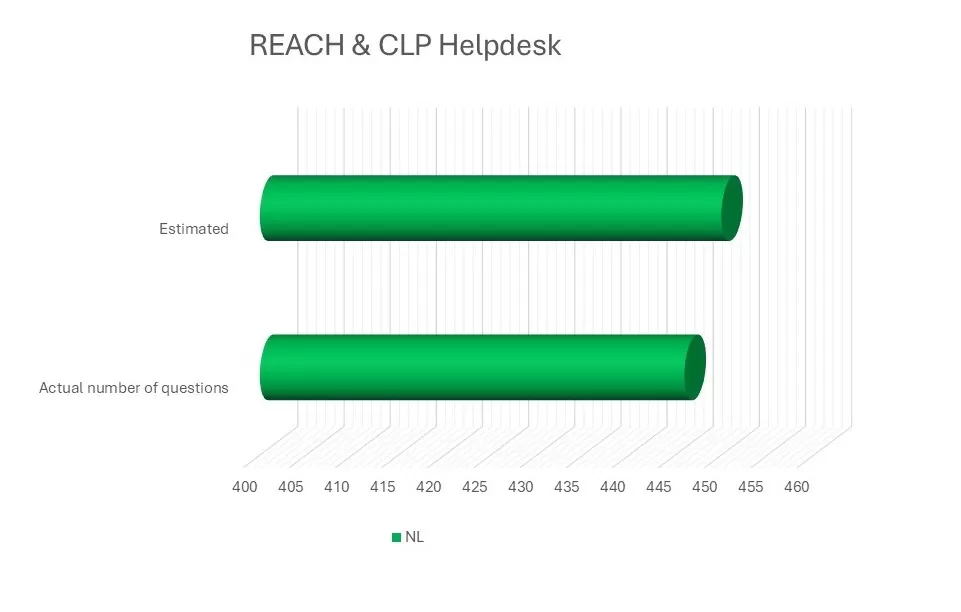
RIVM responds to enquiries submitted to both the REACH Helpdesk and the CLP Helpdesk. The number of enquiries appears to be stabilising in recent years, with 446 questions received and answered.